A unique approach to discovery
MapLight’s multi-pronged discovery platform combines three key technologies that provide us with unique insights into atypical neural circuits. Optogenetics identifies the function of key cell types in the brain. Transcriptomics provides a library of potential therapeutic targets within these cells. STARmap enables us to visualize these targets in neural circuits of intact brain tissue. Together, these tools are driving the development of multiple first-in-class, targeted therapies for difficult-to-treat brain disorders and symptoms.
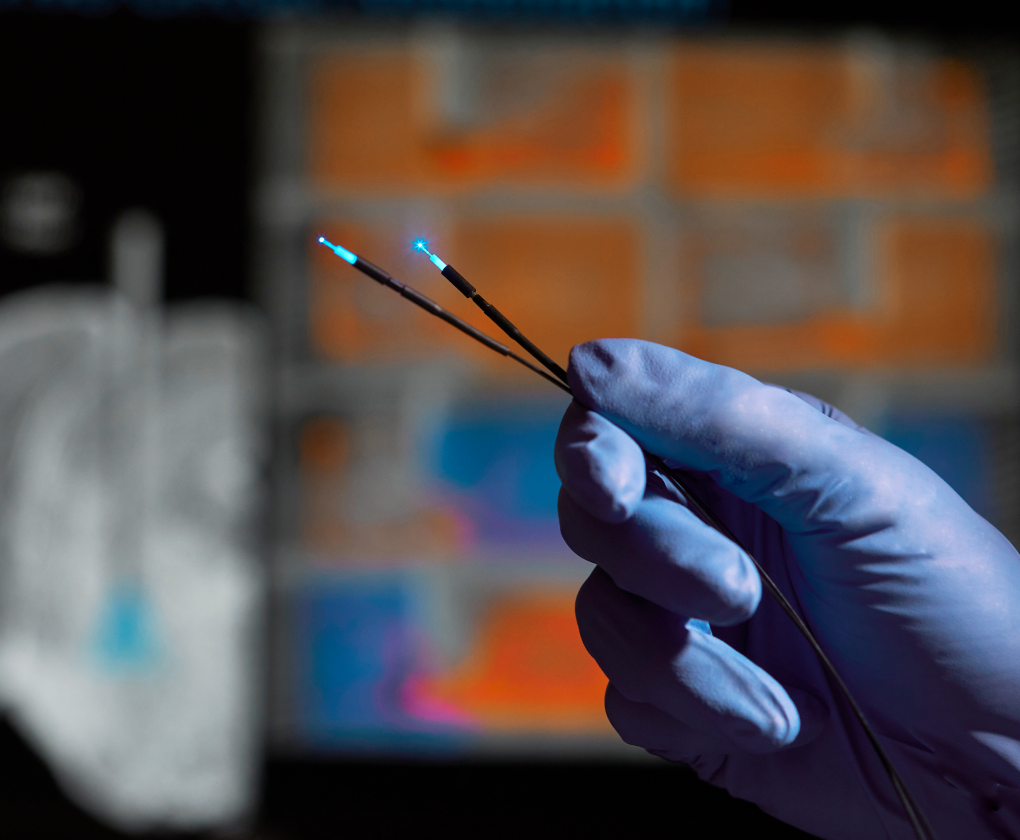
Optogenetics
Optogenetics is a laboratory technique that enables real-time activation and inhibition of the neural circuits that are responsible for the clinical manifestations of central nervous system disorders.
Optogenetics allows us to turn neurons “on” or “off” with the flick of a light switch. We start with opsin proteins that can be activated or deactivated with light– excitatory ion channels such as ChR2 that trigger action potential firing, or inhibitory Cl– pumps such as NpHR that silence neural activity. We then use genetically targeted viruses to specify which cells contain the light sensitive opsins, so that only cells in the circuit of interest will be responsive to the light. The resulting changes in behavior can then be titrated and monitored in real time, illustrating a direct causal link between changes in neural circuit activity and either the emergence or resolution of disease-relevant symptoms.
Circuit function is systematically tested across multiple nodes in a network, and parameters are adjusted to define the full range of symptoms controlled by individual circuits.
Transcriptomics
Transcriptomics is a unique approach for identifying the gene expression pattern of each cell type in the brain, enabling the identification of potential treatment targets within specific brain circuits.
With transcriptomics, gene expression profiling is used to identify gene products capable of modulating different circuit elements and the disease symptoms they mediate. MapLight predicts how selective a drug target is for a given cell type and identifies tool compounds for modulating these targets or performs molecular manipulations to assess how effectively these targets can modulate the pathological circuit activity mediating disease symptoms.
MapLight’s proprietary bioinformatic algorithm can analyze gene expression data from millions of single cells across the central and peripheral nervous systems. This rich resource is used to fuel the identification of novel cell types and targets, to rank candidates by specificity to the circuit of interest, and to predict synergistic drug combinations.

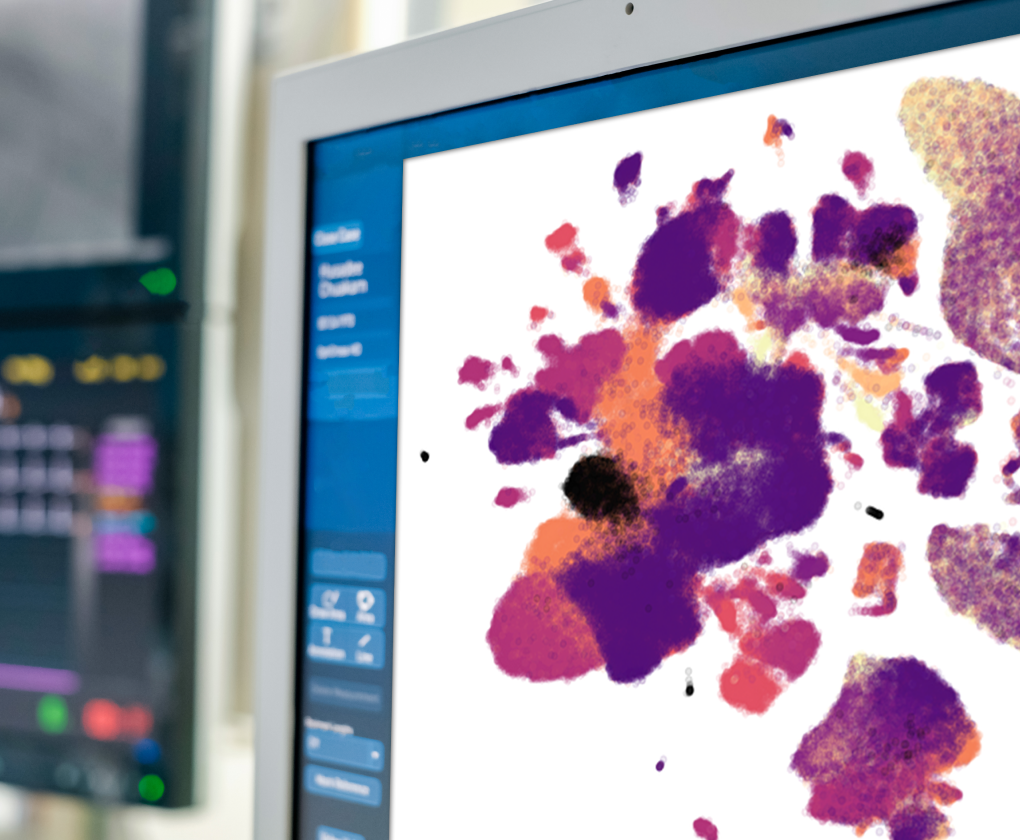
STARmap
STARMap is a patented technology that enables the visualization of potential therapeutic targets in neural circuits within intact brain tissue.
With STARmap technology, hundreds of gene transcripts can be assayed in situ, preserving the spatial integrity and anatomical architecture of the intact brain circuit. This allows us to efficiently locate drug target candidates within key neural circuits and combine this information with functional (e.g. immediate early gene expression) and anatomical (e.g. projection pathway) readouts.
The architecture and genetic composition of circuits can be confirmed across species to prioritize targets most likely to effectively translate from preclinical models into clinical patient populations.
STARmap is a spatial transcriptomics technology that allows for the visualization of gene expression in neural circuits within preserved slices of brain tissue. STARmap data is generated by processing these brain slices to render them transparent and then using fluorescent labels to mark the location of different transcripts in the tissue. STARmap produces a microscopic image of intact brain tissue overlaid with thousands of colored dots, with each color corresponding to a specific drug target of interest. MapLight uses this technology to verify the expression, selectivity and location of druggable targets in different circuits of the brain. STARmap can be applied to brain tissue across species, including humans, which allows MapLight to validate that our drug targets are located within disease-relevant neural circuits in humans.